US Pharmaceutical Market Size, Share, In-Depth Analysis, Opportunity and Forecast Report by 2033
IMARC Group has recently released a new research study titled “US Pharmaceutical Market Size, Share, Trends and Forecast by Prescription Type, Therapeutic Category, and Region, 2025-2033”, offers a detailed analysis of the market drivers, segmentation, growth opportunities, trends and competitive landscape to understand the current and future market scenarios.
US Pharmaceutical Market Overview
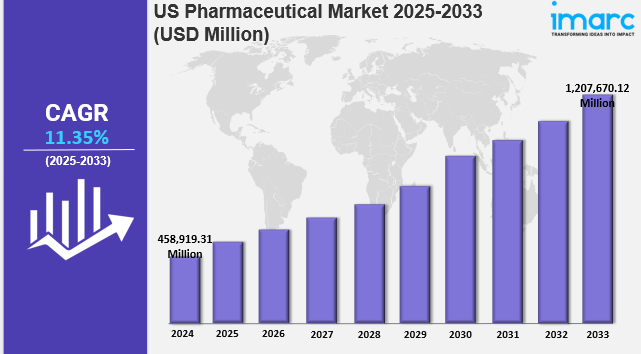
The US pharmaceutical market size reached USD 458,919.31 Million in 2024. The market is projected to reach USD 1,207,670.12 Million by 2033, exhibiting a growth rate (CAGR) of 11.35% during 2025-2033. The pharmaceutical market in US is driven by the rising healthcare needs from an aging population, rapid digital integration transforming research and treatment processes, and expanding partnerships that combine resources and expertise. These forces collectively reinforce innovation, operational effectiveness, and long-term competitiveness across the country pharmaceutical landscape, thereby influencing the US pharmaceutical market share.
Key Market Highlights – United States Pharmaceutical Market
✔ Strong market expansion supported by rising healthcare awareness and an aging population requiring continuous medication management
✔ Increasing prescription volumes driven by higher prevalence of chronic diseases such as diabetes, hypertension, and cardiovascular conditions
✔ Growing consumer preference for convenient pharmacy formats, including retail chains, mail-order services, and online pharmacies
✔ Expansion of clinical services within pharmacies, including vaccination programs, health screenings, and medication therapy management
✔ Rapid digital transformation with e-prescriptions, mobile health apps, and automated dispensing systems enhancing operational efficiency
✔ Rising demand for specialty drugs and biologics, positioning pharmacies as critical distribution and patient support centers
Request for a sample copy of the report:https://www.imarcgroup.com/us-pharmaceutical-market/requestsample
Trends in the US Pharmaceutical Market
Several key trends are emerging in the US pharmaceutical market, reflecting the changing landscape of healthcare and evolving consumer expectations. One prominent trend is the increasing emphasis on patient-centric approaches, where pharmaceutical companies are focusing on improving patient engagement and adherence to therapies.
This shift is leading to the development of digital health solutions, such as mobile apps and telehealth platforms, which facilitate communication between patients and healthcare providers, enhancing the overall treatment experience. Additionally, the US pharmaceutical market is witnessing a rise in biosimilars, as healthcare providers and payers seek cost-effective alternatives to expensive biologic therapies.
The growing acceptance of biosimilars is expected to increase competition, driving down costs and expanding access to essential medications. Furthermore, sustainability is becoming a critical focus for pharmaceutical companies, with many organizations committing to environmentally friendly practices in their operations and supply chains. As the market approaches 2026, these trends indicate a transformative period for the US pharmaceutical market, where patient engagement, cost management, and sustainability will play pivotal roles in shaping the future of healthcare delivery.
Market Dynamics of the US Pharmaceutical Market
Rising Demand for Specialty Drugs
The US pharmaceutical market is witnessing a significant surge in demand for specialty drugs, driven by the increasing prevalence of chronic diseases and complex health conditions. Specialty drugs, which often require special handling, administration, and monitoring, are becoming essential in the treatment of conditions such as cancer, multiple sclerosis, and rheumatoid arthritis.
As the population ages and the incidence of chronic illnesses rises, healthcare providers are increasingly turning to these advanced therapies to improve patient outcomes. This trend is particularly evident in the oncology sector, where innovative therapies such as immunotherapy and targeted treatments are gaining traction.
Additionally, the high cost associated with specialty drugs is prompting payers and providers to explore value-based care models that emphasize efficacy and patient outcomes over traditional volume-based reimbursement. As a result, the US pharmaceutical market is expected to see substantial growth in specialty drug sales, which will significantly impact overall market dynamics and necessitate strategic planning from pharmaceutical companies to manage pricing and access.
Technological Advancements in Drug Development
Technological advancements are revolutionizing the US pharmaceutical market, particularly in drug discovery and development processes. Innovations such as artificial intelligence (AI), machine learning, and big data analytics are enabling pharmaceutical companies to streamline research and development (R&D), reducing the time and cost associated with bringing new drugs to market.
These technologies facilitate the identification of potential drug candidates, optimize clinical trial designs, and enhance patient recruitment efforts, ultimately accelerating the development pipeline. Furthermore, advancements in genomics and biotechnology are paving the way for personalized medicine, allowing for tailored therapies that address the unique genetic makeup of individual patients.
As these technologies continue to evolve, they are expected to drive efficiency and effectiveness in the pharmaceutical R&D process, leading to the introduction of novel therapies that meet the growing demands of the healthcare market. The US pharmaceutical market is likely to benefit significantly from these innovations, as companies leverage technology to enhance their competitive edge and respond to the evolving healthcare landscape.
Regulatory Changes and Market Access Challenges
Regulatory changes and market access challenges are shaping the landscape of the US pharmaceutical market, influencing how companies develop, price, and distribute their products. Recent legislative efforts aimed at reducing drug prices and increasing transparency in pricing practices are prompting pharmaceutical companies to reassess their pricing strategies and market access approaches.
The introduction of policy measures such as the Inflation Reduction Act, which allows Medicare to negotiate prices for certain high-cost drugs, is particularly impactful. These changes are pushing pharmaceutical companies to focus on demonstrating the value of their products through robust clinical evidence and real-world data. Additionally, navigating the complex landscape of payer negotiations and formulary placements is becoming increasingly critical for market success.
As a result, pharmaceutical companies must invest in strategic market access initiatives and engage with stakeholders early in the drug development process to ensure their products achieve optimal reimbursement and patient access. This dynamic environment is expected to drive innovation and adaptability within the US pharmaceutical market.
United States Pharmacy Market Segmentation:
Prescription Type Insights:
- Prescription Drugs
- Branded
- Generics
- OTC Drugs
Therapeutic Category Insights:
- Antiallergics
- Blood and Blood Forming Organs
- Cardiovascular System
- Dermatological
- Genito Urinary System
- Respiratory System
- Sensory Organs
- Others
Regional Insights:
- Northeast
- Midwest
- South
- West
Competitive Landscape:
The market research report has also provided a comprehensive analysis of the competitive landscape. Competitive analysis such as market structure, key player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided.
Speak to An Analyst:https://www.imarcgroup.com/request?type=report&id=43452&flag=C
US Pharmaceutical Market News:
- October 2025: Lupin launched a generic version of Janssen’s Xarelto (Rivaroxaban for Oral Suspension, 1 mg/mL) in the US. The drug treats and prevents recurrent venous thromboembolism (VTE) in pediatric patients.
- August 2025: Natco Pharma launched Bosentan tablets in the US for treating pulmonary arterial hypertension in children aged 3 and above. The drug, a generic version of Tracleer, comes with 180-day exclusivity due to Natco’s first-to-file status. Lupin Ltd is Natco’s marketing partner, and the drug saw estimated US sales of USD 10 million over the past year.
About Us
IMARC Group is a global management consulting firm that helps the world’s most ambitious changemakers to create a lasting impact. The company provide a comprehensive suite of market entry and expansion services. IMARC offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us
IMARC Group,
134 N 4th St. Brooklyn, NY 11249, USA,
Email: sales@imarcgroup.com,
Tel No: (D) +91 120 433 0800,
United States: +1-201971-6302
- Business
- Research
- Energy
- Art
- Causes
- Tech
- Crafts
- crypto
- Dance
- Drinks
- Film
- Fitness
- Food
- Spiele
- Gardening
- Health
- Startseite
- Literature
- Music
- Networking
- Andere
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness



